| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 5mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| Other Sizes |
|
| 靶点 |
AMPA (IC50 = 0.3 μM); kainate receptor (IC50 = 1.5 μM)[1]
AMPA/Kainate receptors (non-NMDA subtype of glutamate receptors). The study demonstrates a clear preference of CNQX for non-NMDA receptors. [1] Non-NMDA type glutamate receptors, with higher affinity for the quisqualate receptor subtype compared to NMDA and kainate receptor subtypes (as referenced from binding studies). [2] Non-NMDA subtype of excitatory amino acid receptors (specifically kainate and quisqualate/AMPA receptors). The study confirms its selectivity against NMDA receptors. [3] CNQX is an antagonist of the AMPA/kainate subtype of ionotropic glutamate receptors. The text also notes that compounds in the quinoxalinedione series, including CNQX, have inhibitory activity at kainate receptors and have been reported to show affinity for the glycine site of the NMDA receptor. [4] |
|---|---|
| 体外研究 (In Vitro) |
CNQX (FG9065; 2–5 μM) 避免快速和慢速 GABA 介导的海马切片灌注抑制,同时可逆地扩张 Schaffer 侧枝和丝状兴奋性突触后电位 (EPSP) [2]。当 CNQX (1-5 μM) 接地时,从具有舞蹈兴奋的腰节记录的 DR-VRR 单突触成分的幅度会降低 [3]。
在大鼠脊髓神经元的在体电生理记录中,电泳给予CNQX(从1 mM溶液中电泳释放)能大致同等程度地降低激动剂使君子酸和红藻氨酸诱导的兴奋性反应。例如,在一个神经元中,CNQX (20 nA) 降低了由使君子酸和红藻氨酸引起的放电频率增加。该效应起效和恢复迅速。相比之下,CNQX对N-甲基-DL-天冬氨酸(NMA,一种NMDA受体激动剂)诱导的兴奋反应几乎没有影响。研究指出,在降低使君子酸和红藻氨酸反应方面,CNQX的效力明显高于另一种化合物GAMS。[1] 讨论中引用的受体结合实验指出,与在[3H]AMPA结合分析中的抑制活性相比,CNQX对[3H]红藻氨酸结合(特别是在Ca2+敏感的高亲和力位点)的抑制作用相对较弱。这表明其在生理条件下的功能性拮抗作用可能主要涉及使君子酸(AMPA)受体。[1] 在大鼠海马脑片制备中,灌注CNQX (2-5 µM) 可完全且可逆地阻断CA1区谢弗侧支通路和CA3区苔藓纤维通路刺激诱发的兴奋性突触后电位。阻断作用迅速(4 µM CNQX约2分钟),恢复需要长达30分钟的冲洗。CNQX的突触阻断作用与记录神经元的膜电位、输入电阻或锋电位适应性的改变无关。[2] CNQX (2-4 µM) 在消除突触兴奋的同时,不改变由GABA介导的快和慢抑制性突触后电位的幅度,表明其不影响前馈抑制通路。[2] 在CA3神经元的电压钳实验中,CNQX 在降低激动剂诱导的内向电流方面表现出选择性。使君子酸 (10 µM) 诱导的电流被4 µM CNQX降低至对照的56 ± 17%,被10 µM CNQX降低至32 ± 4%。NMDA (20 µM) 诱导的电流降低程度较小(暴露5分钟后,分别被4 µM和10 µM CNQX降低至对照的77 ± 14%和66 ± 11%)。红藻氨酸 (200 nM) 诱导的内向电流被10 µM CNQX轻微降低(降至对照的78 ± 17%)。相比之下,NMDA受体拮抗剂AP-5或AP-7 (50 µM) 可降低NMDA诱导的电流,但不影响使君子酸或红藻氨酸诱导的电流。CNQX对测试激动剂的拮抗效力顺序为使君子酸 > 红藻氨酸 = NMDA。[2] 在离体七鳃鳗脊髓制备中,浴槽施加CNQX (5 µM) 阻断了运动神经元中记录的复合单突触兴奋性突触后电位的快速、非NMDA受体介导成分。在含有生理浓度Mg²⁺的正常林格氏液中应用时,CNQX留下了一个较小、较慢的EPSP成分,该成分随后可被NMDA受体拮抗剂AP5阻断,这表明CNQX对非NMDA受体具有选择性。[3] CNQX (剂量高达15 µM) 对由浴槽应用NMDA受体激动剂N-甲基-D,L-天冬氨酸 (NMA, 100 µM) 诱导的虚拟运动(一种运动模式)的节律或组织没有影响。[3] 在TTX和低Mg²⁺存在下,将激动剂压力注射到运动神经元上的实验显示,浴槽应用DNQX (5 µM) 阻断了了对红藻氨酸和AMPA的反应,但不影响对NMDA的反应。虽然在这个特定图表/实验中未直接测试CNQX,但文中将CNQX与DNQX归为一类,认为它们在阻断非NMDA突触传递方面具有相同的特异性。[3] |
| 体内研究 (In Vivo) |
CNQX (FG9065; 2–5 μM) 避免快速和慢速 GABA 介导的海马切片灌注抑制,同时可逆地扩张 Schaffer 侧枝和丝状兴奋性突触后电位 (EPSP) [2]。通过配音从腰节记录的 DR-VRR 单突触分量的幅度大约被 CNQX (1 -5 μM) 降低[3]。
在无可卡因阶段的前 15 分钟,CNQX 二钠(FG9065 二钠;0.75–3 mg/kg;腹腔注射;测试前 20 分钟)剂量依赖性地减少可卡因反应的次数 [4]。保留试验前10分钟,可将CNQX二钠(0.5或1.25μg)双侧注入杏仁核或背侧海马,以部分降低训练后24小时大鼠低血压抑制性回避的表达。 0.5 μg 时,CNQX 二钠完全阻断通道 [5]。 七鳃鳗运动的运动模式是由兴奋性氨基酸神经传递激活和维持的。喹喔啉二酮6,7-二硝基喹喔啉-2,3-二酮(DNQX)和6-氰基-7-硝基喹喔啉-2,3-二酮(CNQX)是哺乳动物中枢神经系统中非N-甲基-D-天冬氨酸(NMDA)受体的强效选择性拮抗剂。在七鳃鳗中,这些化合物现在被证明可以阻断脊髓腹角神经元中诱发的快速兴奋性突触电位。它们选择性地拮抗对选择性红藻氨酸和奎司琼受体激动剂(红藻氨酸及α-氨基-3-羟基-5-甲基-4-异恶酮(AMPA))应用的反应,但不影响NMDA受体介导的反应。此外,研究表明,在用DNQX或CNQX阻断非NMDA受体后,NMDA受体的激活足以引发和维持假想运动。相反,用AMPA激活奎司特受体,而不是奎司特,会导致具有与红藻氨酸激活的特性非常相似的虚构运动[3]。 增加可卡因单位剂量在第一和第二间隔期间增加了反应,第一次CS的潜伏期缩短。根据FR4(FR7:S)时间表,CNQX在前15分钟无可卡因间隔期间以剂量依赖的方式减少了可卡因反应的数量,但在第二个间隔或会话的后半部分都没有影响可卡因反应。在运动活动测试中,与在第一个固定间隔内显著减弱反应的剂量相比,更高的CNQX剂量会减少饲养。 结论:这些结果表明,AMPA/红藻氨酸受体参与了可卡因寻求行为的介导,部分受可卡因相关线索的控制[4]。 使用恐惧强化惊吓范式研究了杏仁核中非N-甲基-D-天冬氨酸受体在条件性恐惧表达中的作用。在杏仁核基底外侧核植入双侧套管的大鼠在每2天接受10对视觉或听觉条件刺激,并伴有足部电击。第二天,他们在有或没有条件刺激的情况下通过诱发声惊吓反射进行测试,并分为具有同等增强水平的组。一两天后,在杏仁核内输注赋形剂或0.025、0.25或2.5微克6-氰基-7-硝基喹喔啉-2,3-二酮后,再次对大鼠进行测试。该药物剂量依赖性地阻断了两种感觉模式中强化惊吓的表达,表明杏仁核中非NMDA受体的激活对于条件性恐惧的表达是必要的[5]。 1. 全身性腹膜内注射 AMPA/红藻氨酸受体拮抗剂 CNQX(0.75、1.5 和 3 mg/kg)能剂量依赖性地减少大鼠在可卡因强化二阶程序下,第一个无药固定间隔期内的有效杠杆按压次数。这种抑制作用特定于初始的“可卡因寻求”阶段,因为在第一次可卡因输注后的第二个间隔期内的反应以及后续会话中自我给药的可卡因输注总次数并未受到影响。[4] 2. 在单独的自发活动性测试中,3 mg/kg IP 剂量的 CNQX 显著抑制了垂直活动( rearing),但并未减少水平活动;事实上,它在特定时间段内增加了水平活动。3 mg/kg 剂量对可卡因寻求的减弱可能部分与运动功能损害效应有关,但能够减弱寻求行为的较低剂量并未显著影响 rearing。[4] |
| 酶活实验 |
海马脑片与6-氰基-7-硝基喹喔啉-2,3-二酮(CNQX,2-5微M)的超融合可逆地阻断了Schaffer侧支和苔藓纤维兴奋性突触后电位(EPSP),同时保留了快速和慢速γ-氨基丁酸(GABA)介导的抑制。膜电位、输入电阻和刺突调节没有改变。与红藻氨酸或N-甲基-D-天冬氨酸诱导的电流相比,CNQX在更大程度上减少了奎奎司特诱导的内向电流。我们认为CNQX可能是研究兴奋性氨基酸介导的突触传递的有用拮抗剂[2]。
|
| 动物实验 |
动物/疾病模型:雄性Wistar大鼠,体重180-200 g[4] 剂量:0.75、1.5和3 mg/kg
给药途径:腹腔注射;测试前20分钟 实验结果:在前15分钟无可卡因给药期间,可卡因(静脉注射;0.25 mg/次)反应次数呈剂量依赖性降低 电生理实验在戊巴比妥麻醉的大鼠上进行。使用填充有3.6M NaCl溶液的七管微电极的中心管记录脊髓(背角)单个神经元的动作电位。微电极的外管包含用于电泳(离子导入)的药物溶液。定位神经元后,通过周期性注射激动剂奎斯奎酸、红藻氨酸和NMA来建立基线兴奋性反应。将CNQX溶解于浓度为1 mM的200 mM CaCl2溶液(pH值调至9.8)中,以便加载到微电极管中。然后使用电泳电流(例如20 nA)将其注射到记录神经元附近,以研究其对激动剂诱导的神经元放电率的影响。[1] 从雄性Wistar大鼠(90-125 g)制备横向海马切片(厚度500 µm)。将切片转移到浸没式记录室中,并以33-34°C的氧合人工脑脊液(ACSF)持续灌注(2 ml/min)。人工脑脊液(ACSF)的成分(单位:mM)为:NaCl 126、KCl 3.5、MgCl₂ 1.3、NaH₂PO₄ 1.2、CaCl₂ 2、NaHCO₃ 25 和葡萄糖 11,并用 95% O₂/5% CO₂ 混合气体通气至 pH 7.3。细胞内记录和电压钳实验均使用微电极。CNQX 通过灌流方式施加,浓度范围为 2 至 10 µM。在一些电压钳实验中,使用河豚毒素(TTX,1 µM)阻断电压门控钠通道,并在微电极中填充 3 M CsCl 以降低钾电流。[2] 实验对象为成年河七鳃鳗(Lampetra fluviatilis)。进行细胞内记录和突触传递研究时,脊髓被完全分离。为了研究模拟运动,将脊髓片段保留在下方的脊索上。将标本浸泡在冷却的(8-10°C)七鳃鳗林格氏液中。在一些细胞内记录实验中,溶液中不含Mg²⁺。在涉及激动剂喷射的实验中,将药物(例如,NMDA、红藻氨酸、AMPA)通过置于脊髓上方的微电极进行压力喷射。对于浴液给药,将CNQX、DNQX、NMA或AMPA等药物以特定浓度(例如,CNQX 5-15 µM)直接添加到浴液林格氏液中。使用吸附电极记录腹根活动以监测模拟运动。[3]可卡因自我给药和CNQX测试:雄性Wistar大鼠接受训练,在复杂的二级强化程序[FI15 min(FR7:S)]下进行静脉注射可卡因的自我给药,随后进行一段时间的FR4(FR7:S)强化程序训练。在0.50 mg/次的可卡因剂量下建立稳定的反应后,测试CNQX的作用。将CNQX(0、0.75、1.5、3 mg/kg)溶于蒸馏水中,并在自我给药实验开始前20分钟以1 ml/kg的体积进行腹腔注射(IP)。剂量以平衡顺序(拉丁方设计)给药,每周一次。[4] 2. 运动活性测试:注射载体后,大鼠适应测试笼。注射CNQX(0、0.75、1.5、3 mg/kg,腹腔注射)20分钟后,检测其作用。记录60分钟的运动活性(通过光电管光束检测水平和垂直运动)。实验采用受试者内拉丁方设计,不同剂量CNQX的测试之间至少间隔两天的载体注射。[4] 3. 手术操作:在异氟烷麻醉下,将慢性颈静脉导管植入大鼠体内,用于静脉给药。每日用抗生素和肝素化生理盐水冲洗导管,以保持导管通畅并预防感染。[4] |
| 参考文献 |
|
| 其他信息 |
6-氰基-7-硝基喹喔啉-2,3-二酮是一种喹喔啉衍生物。
它是一种强效的兴奋性氨基酸拮抗剂,对非NMDA型离子型受体具有选择性。它主要用作研究工具。 CNQX(6-氰基-7-硝基喹喔啉-2,3-二酮)在本研究中被鉴定为一种强效且选择性的非NMDA型谷氨酸受体(特别是奎斯奎酸/AMPA和红藻氨酸受体)拮抗剂,对NMDA受体介导的反应影响极小。它被用作药理学工具,以解析不同兴奋性氨基酸受体亚型在哺乳动物脊髓突触传递中的作用。 CNQX 和 DNQX 的开发为研究奎斯奎酸和红藻氨酸受体亚型开辟了新的途径。[1] 本研究将 CNQX(6-氰基-7-硝基喹喔啉-2,3-二酮)表征为海马兴奋性突触传递的强效、选择性和可逆性拮抗剂,该传递由非 NMDA 谷氨酸受体介导。CNQX 能够阻断突触 EPSP,同时不影响 GABA 能抑制,并且其对奎斯奎酸诱导电流的拮抗作用优于 NMDA 诱导电流,这些特性表明 CNQX 是研究兴奋性氨基酸介导的突触传递的有效药理学工具。研究结果支持了以下假设:Schaffer侧支和苔藓纤维EPSP的神经递质主要作用于奎斯奎酸型受体。[2] 这项在七鳃鳗脊髓中进行的研究证实,CNQX是一种强效且选择性的非NMDA(红藻氨酸和奎斯奎酸/AMPA)兴奋性氨基酸受体介导的突触传递拮抗剂,这与其在哺乳动物中枢神经系统中的作用一致。一项关键发现是,用CNQX阻断非NMDA受体并不会破坏仅由NMDA受体激活诱导的“虚拟运动”(游泳的协调运动模式),这表明NMDA受体介导的突触传递足以协调这种节律性运动活动。该研究还指出,与浓度极高(>10 µM)的 DNQX 不同,浓度高达 15 µM 的 CNQX 不会干扰 NMDA 诱发的运动,这表明在所用剂量下 CNQX 具有更纯净的药理学特性,这可能是由于其对 NMDA 受体复合物甘氨酸位点的作用较小。[3] 1. CNQX(6-氰基-7-硝基喹喔啉-2,3-二酮二钠)是一种水溶性 AMPA/红藻氨酸受体拮抗剂。[4] 2. 该研究表明,AMPA/红藻氨酸受体参与介导可卡因渴求行为,而这种行为至少部分受可卡因相关环境线索(条件刺激)的控制。系统性CNQX的作用仅限于可卡因摄入前的线索控制的“寻求”阶段,而非可卡因输注后的“服用”阶段。[4] 3. 该文章讨论了可能涉及的神经回路,包括从前扣带回皮层和基底外侧杏仁核到伏隔核核心的谷氨酸能投射。[4] |
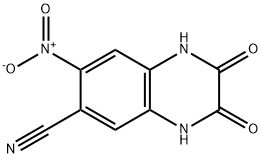
| 分子式 |
C₉H₄N₄O₄
|
|---|---|
| 分子量 |
232.15
|
| 精确质量 |
232.023
|
| CAS号 |
115066-14-3
|
| 相关CAS号 |
CNQX disodium;479347-85-8
|
| PubChem CID |
3721046
|
| 外观&性状 |
White to yellow solid powder
|
| 密度 |
1.7±0.1 g/cm3
|
| 沸点 |
659.3ºC at 760 mmHg
|
| 熔点 |
300 °C
|
| 闪点 |
352.6ºC
|
| 蒸汽压 |
0.001mmHg at 25°C
|
| 折射率 |
1.699
|
| LogP |
0.64
|
| tPSA |
135.33
|
| 氢键供体(HBD)数目 |
2
|
| 氢键受体(HBA)数目 |
5
|
| 可旋转键数目(RBC) |
0
|
| 重原子数目 |
17
|
| 分子复杂度/Complexity |
434
|
| 定义原子立体中心数目 |
0
|
| InChi Key |
RPXVIAFEQBNEAX-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C9H4N4O4/c10-3-4-1-5-6(2-7(4)13(16)17)12-9(15)8(14)11-5/h1-2H,(H,11,14)(H,12,15)
|
| 化学名 |
7-nitro-2,3-dioxo-1,4-dihydroquinoxaline-6-carbonitrile
|
| 别名 |
FG9065; 6-CYANO-7-NITROQUINOXALINE-2,3-DIONE; 7-Nitro-2,3-dioxo-1,2,3,4-tetrahydroquinoxaline-6-carbonitrile; 7-nitro-2,3-dioxo-1,4-dihydroquinoxaline-6-carbonitrile; 6-Quinoxalinecarbonitrile, 1,2,3,4-tetrahydro-7-nitro-2,3-dioxo-; FG 9065; FG-9065;FG 9065
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
DMSO : ~20 mg/mL (~86.15 mM)
H2O : < 0.1 mg/mL |
|---|---|
| 溶解度 (体内实验) |
注意: 如下所列的是一些常用的体内动物实验溶解配方,主要用于溶解难溶或不溶于水的产品(水溶度<1 mg/mL)。 建议您先取少量样品进行尝试,如该配方可行,再根据实验需求增加样品量。
注射用配方
注射用配方1: DMSO : Tween 80: Saline = 10 : 5 : 85 (如: 100 μL DMSO → 50 μL Tween 80 → 850 μL Saline)(IP/IV/IM/SC等) *生理盐水/Saline的制备:将0.9g氯化钠/NaCl溶解在100 mL ddH ₂ O中,得到澄清溶液。 注射用配方 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (如: 100 μL DMSO → 400 μL PEG300 → 50 μL Tween 80 → 450 μL Saline) 注射用配方 3: DMSO : Corn oil = 10 : 90 (如: 100 μL DMSO → 900 μL Corn oil) 示例: 以注射用配方 3 (DMSO : Corn oil = 10 : 90) 为例说明, 如果要配制 1 mL 2.5 mg/mL的工作液, 您可以取 100 μL 25 mg/mL 澄清的 DMSO 储备液,加到 900 μL Corn oil/玉米油中, 混合均匀。 View More
注射用配方 4: DMSO : 20% SBE-β-CD in Saline = 10 : 90 [如:100 μL DMSO → 900 μL (20% SBE-β-CD in Saline)] 口服配方
口服配方 1: 悬浮于0.5% CMC Na (羧甲基纤维素钠) 口服配方 2: 悬浮于0.5% Carboxymethyl cellulose (羧甲基纤维素) 示例: 以口服配方 1 (悬浮于 0.5% CMC Na)为例说明, 如果要配制 100 mL 2.5 mg/mL 的工作液, 您可以先取0.5g CMC Na并将其溶解于100mL ddH2O中,得到0.5%CMC-Na澄清溶液;然后将250 mg待测化合物加到100 mL前述 0.5%CMC Na溶液中,得到悬浮液。 View More
口服配方 3: 溶解于 PEG400 (聚乙二醇400) 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.3076 mL | 21.5378 mL | 43.0756 mL | |
| 5 mM | 0.8615 mL | 4.3076 mL | 8.6151 mL | |
| 10 mM | 0.4308 mL | 2.1538 mL | 4.3076 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。