| 规格 | 价格 | |
|---|---|---|
| 500mg | ||
| 1g | ||
| Other Sizes |
| 靶点 |
DPP-4
|
|---|---|
| 药代性质 (ADME/PK) |
Absorption
In healthy subjects, after a single oral dose of 5 mg saxagliptin, the mean plasma AUC values of saxagliptin and its active metabolite were 78 ng·h/mL and 214 ng·h/mL, respectively. The corresponding plasma Cmax values were 24 ng/mL and 47 ng/mL, respectively. No accumulation of saxagliptin occurred after repeated dosing. After a once-daily dose of 5 mg, the median time to peak concentration (Tmax) of saxagliptin was 2 hours, and the median time to peak concentration of its active metabolite was 4 hours. Bioavailability (2.5–50 mg dose) = 67% Elimination Pathway Saxagliptin is primarily eliminated via the renal and hepatic routes. After a single dose of 50 mg 14C-saxagliptin, 24%, 36%, and 75% of the dose were excreted in the urine as saxagliptin, its active metabolite, and total radioactivity, respectively. 22% of the administered radioactivity was recovered in feces, representing the dose of saxagliptin excreted in bile and/or the drug not absorbed from the gastrointestinal tract. Volume of distribution 151 L Clearance Renal clearance, single 50 mg dose = 14 L/h A single-dose, open-label study aimed to evaluate the pharmacokinetics of saxagliptin (10 mg dose) in subjects with varying degrees of chronic renal impairment (8 patients per group) and subjects with normal renal function. The 10 mg dose is not the approved dose. The study included patients with renal impairment categorized by creatinine clearance as mild (>50 to ≥80 mL/min), moderate (30 to ≥50 mL/min), and severe (<30 mL/min), as well as patients with end-stage renal disease undergoing hemodialysis. …The degree of renal impairment did not affect the Cmax of saxagliptin and its active metabolites. In patients with mild renal impairment, the AUC values of saxagliptin and its active metabolite were 20% and 70% higher, respectively, than in patients with normal renal function. Since this increase is not clinically significant, dose adjustment is not recommended for patients with mild renal impairment. In patients with moderate or severe renal impairment, the AUC values of saxagliptin and its active metabolite were 2.1 times and 4.5 times higher, respectively, than in patients with normal renal function. To ensure similar plasma exposure of saxagliptin and its active metabolite to patients with normal renal function, the recommended dose for patients with moderate to severe renal impairment and end-stage renal disease requiring hemodialysis is 2.5 mg once daily. Saxagliptin is removed by hemodialysis. View MoreSaxagliptin is cleared via the kidneys and liver. Following a single oral dose of 50 mg (14)-C-saxagliptin, 24%, 36%, and 75% of the dose were excreted in the urine as saxagliptin, its active metabolites, and total radioactivity, respectively. The mean renal clearance of saxagliptin (approximately 230 mL/min) was greater than the mean estimated glomerular filtration rate (approximately 120 mL/min), suggesting partial active renal excretion. The total radioactivity recovered in the feces was 22% of the administered dose, representing the dose of saxagliptin excreted via bile and/or not absorbed from the gastrointestinal tract. Saxagliptin is rapidly absorbed after oral administration on an empty stomach, with peak plasma concentrations (Cmax) of saxagliptin and its major metabolite reached within 2 and 4 hours, respectively. The Cmax and AUC values of saxagliptin and its major metabolite increase proportionally with increasing saxagliptin dose, and this dose-proportionality is still observed at doses up to 400 mg. In healthy subjects, after a single oral dose of 5 mg saxagliptin, the mean plasma AUC values of saxagliptin and its major metabolite were 78 nghr/mL and 214 nghr/mL, respectively, with corresponding plasma Cmax values of 24 ng/mL and 47 ng/mL. The intra-subject coefficients of variation for both saxagliptin Cmax and AUC were less than 12%. Metabolism/Metabolites The metabolism of saxagliptin is primarily mediated by cytochrome P450 3A4/5 (CYP3A4/5). 50% of the absorbed dose is metabolized by the liver. Saxagliptin's major metabolite, 5-hydroxysaxagliptin, is also a DPP4 inhibitor, with approximately half the potency of saxagliptin. The metabolism of saxagliptin is primarily mediated by CYP3A4/5. In vitro studies have shown that saxagliptin and its active metabolite do not inhibit CYP1A2, 2A6, 2B6, 2C9, 2C19, 2D6, 2E1, or 3A4, nor do they induce CYP1A2, 2B6, 2C9, or 3A4. Therefore, saxagliptin is not expected to alter the metabolic clearance of drugs co-metabolized with these enzymes. Saxagliptin is a substrate of P-glycoprotein (P-gp), but not a significant inhibitor or inducer of P-gp. The major metabolite of saxagliptin is also a DPP4 inhibitor, with approximately half the potency of saxagliptin. Biological Half-Life Saxagliptin = 2.5 hours; 5-Hydroxysaxagliptin = 3.1 hours; Following a single oral dose of 5 mg Onglyza in healthy subjects, the mean plasma terminal half-lives of saxagliptin and its active metabolite were 2.5 hours and 3.1 hours, respectively. |
| 毒性/毒理 (Toxicokinetics/TK) |
Toxicity Overview
Identification and Use: Saxagliptin is a dipeptidyl peptidase-4 (DPP-4) inhibitor used to treat type 2 diabetes. It has been shown in various clinical settings to improve glycemic control in adults with type 2 diabetes as an adjunct to diet and exercise. Human Exposure and Toxicity: Saxagliptin treatment significantly improves glycated hemoglobin (A1C) levels compared to placebo. Cases of overdose have been reported, but most were accidental. Most adults and children/adolescents taking gliptins can be safely managed at home without hospitalization upon assessment at a healthcare facility. Cases of adults intentionally self-harming from taking gliptins have been treated at healthcare facilities, but even at doses up to 18 times the adult treatment dose, hospitalization or serious complications are rare. In healthy subjects, daily administration of up to 400 mg of saxagliptin for 2 weeks, equivalent to 80 times the maximum recommended human dose (MRHD), did not result in any dose-related adverse clinical events or clinically significant effects on corrected QT interval (QTc) or heart rate. Animal studies: Saxagliptin caused adverse skin reactions on the extremities of cynomolgus monkeys (scabs and/or ulcers on the tail, toes, scrotum, and/or nose). At doses up to 20 times the MRHD, skin damage was reversible, but at higher doses, irreversible necrosis occurred in some cases. In developmental studies, higher doses of saxagliptin that caused maternal toxicity also increased fetal uptake (approximately 2069 times and 6138 times the MRHD). Additional effects on estrous cycles, fertility, ovulation, and implantation were observed at doses approximately 6138 times the MRHD. In in vitro Ames bacterial assays, in vitro primary human lymphocyte cytogenetics assays, in vivo rat oral micronucleus assays, in vivo rat oral DNA repair studies, and rat peripheral blood lymphocyte oral in vivo/in vitro cytogenetics studies, saxagliptin, regardless of metabolic activation, did not exhibit mutagenicity or chromosome breakage. The active metabolite did not show mutagenicity in the in vitro Ames bacterial assay. Hepatotoxicity In large clinical trials, the rate of serum enzyme elevation was similar in the saxagliptin treatment groups ( Probability score: E (unproven but suspected rare cause of clinically significant liver injury)). Pregnancy and Lactation Effects ◉ Overview of Use During Lactation There is currently no information on the clinical use of saxagliptin during lactation. Saxagliptin has a shorter half-life than other dipeptidyl peptidase IV inhibitors, therefore it may be a better option among drugs in this class for breastfeeding women. Monitoring of blood glucose levels in breastfed infants is recommended during maternal treatment with saxagliptin. [1] However, especially in breastfed newborns or preterm infants, other medications may be preferred. ◉ Effects on breastfed infants No relevant published literature was found as of the revision date. ◉ Effects on breastfeeding and breast milk No relevant published information was found as of the revision date. View moreInteractions Protein Binding The in vitro protein binding rate of saxagliptin and its active metabolites in human serum is negligible (<10%). |
| 其他信息 |
Saxagliptin is a monocarboxylic acid amide formed by the condensation of the carboxyl group of (2S)-amino(3-hydroxyadamantane-1-yl)acetic acid with the amino group of (1S,3S,5S)-2-azabicyclo[3.1.0]hexane-3-nitriles. It is used in its monohydrate form to treat type 2 diabetes. It is a hypoglycemic agent and an EC 3.4.14.5 (dipeptidyl peptidase IV) inhibitor. It belongs to the adamantane, nitrile, azabicycloalkanes, tertiary alcohols, and monocarboxylic acid amides. Saxagliptin (rINN) is an orally effective hypoglycemic agent (antidiabetic drug) belonging to the novel dipeptidyl peptidase-4 (DPP-4) inhibitor class. It was approved by the FDA on July 31, 2009. Anhydrous saxagliptin is a dipeptidyl peptidase 4 inhibitor. The mechanism of action of anhydrous saxagliptin is as a dipeptidyl peptidase 4 inhibitor. Saxagliptin is a dipeptidyl peptidase-4 (DPP-4) inhibitor that can be used alone or in combination with other oral hypoglycemic agents to treat type 2 diabetes, in conjunction with diet and exercise. Saxagliptin is a relatively new drug, and no clinically significant liver injury has been found to be associated with it. Saxagliptin is a potent, selective, competitive cyanopyrrolidine dipeptidyl peptidase-4 (DPP-4) inhibitor with high oral bioavailability and hypoglycemic activity. Saxagliptin is metabolized into a less active monohydroxy metabolite. Anhydrous saxagliptin is the anhydrous form of saxagliptin, a potent, selective, competitive cyanopyrrolidine dipeptidyl peptidase-4 (DPP-4) inhibitor with high oral bioavailability and hypoglycemic activity. Saxagliptin is metabolized into a less active monohydroxy metabolite. Drug Indications: For the treatment of type 2 diabetes to improve glycemic control, either in combination with other drugs or as monotherapy.
Additional combination therapy with Amlinza is indicated for adult patients aged 18 years and older with type 2 diabetes to improve glycemic control: As monotherapy: for patients whose glycemic control is inadequate with diet and exercise alone and who are unsuitable for metformin due to contraindications or intolerance; As dual oral therapy: when metformin alone, combined with diet and exercise, fails to adequately control glycemic control; when sulfonylureas alone, combined with diet and exercise, fail to adequately control glycemic control and are unsuitable for metformin; when thiazolidinediones alone, combined with diet and exercise, fail to adequately control glycemic control and are suitable for thiazolidinediones; As triple oral therapy: when metformin combined with sulfonylureas alone, combined with diet and exercise, fails to adequately control glycemic control; When diet and exercise alone are insufficient to adequately control blood glucose, it can be used in combination with insulin (with or without metformin). View MoreTherapeutic Use Drug Warnings /Black Box Warning/ Warning: Lactic acidosis. Lactic acidosis is a rare but serious complication that can be caused by metformin accumulation. Septicemia, dehydration, excessive alcohol consumption, liver dysfunction, kidney dysfunction, and acute congestive heart failure can increase the risk. Lactic acidosis often has an insidious onset, with only nonspecific symptoms such as malaise, myalgia, dyspnea, increased drowsiness, and nonspecific abdominal discomfort. Abnormal laboratory findings include decreased pH, increased anion gap, and elevated blood lactate levels. If acidosis is suspected, Amlinza extended-release tablets should be discontinued immediately, and the patient should be taken to a hospital. /Saxagliptin and metformin hydrochloride combination preparations/ The U.S. Food and Drug Administration (FDA) is evaluating new, unpublished findings from a group of academic researchers that suggest an increased risk of pancreatitis and a precancerous cellular lesion called pancreatic duct metaplasia in patients with type 2 diabetes treated with a class of drugs called incretin analogs. These findings are based on examination of pancreatic tissue samples from a small number of post-mortem patients. The FDA has requested that researchers provide methods for collecting and studying these samples, as well as tissue samples, so that the FDA can further investigate the potential pancreatic toxicity associated with incretin analogs. Incretin analogues include exenatide (Byetta, Baidu Ruian), liraglutide (Vituzar), sitagliptin (Jenova, Genomex, Genomex Extended Release, Uvitine), saxagliptin (Amligiz, Combigliza Extended Release), alogliptin (Nesina, Kazzano, Osenib), and linagliptin (Trajeta, Gentaduto). These drugs work by mimicking the body's naturally produced incretin hormones, stimulating the release of insulin after meals. They are used in conjunction with diet and exercise to lower blood sugar in adults with type 2 diabetes. The FDA has not yet reached any new conclusions regarding the safety risks of incretin analogues. This preliminary notification is intended only to inform the public and healthcare professionals, and the FDA plans to obtain and evaluate this new information. …The FDA will release its final conclusions and recommendations after completing its review or obtaining more information. The "Warnings and Precautions" section of the drug labels and patient guides for incretin analogues contains warnings about the risk of acute pancreatitis. The FDA has not previously issued any announcements regarding the risk that incretin analogues may cause precancerous lesions of the pancreas. The FDA has also not concluded that these drugs may cause or promote pancreatic cancer. Currently, patients should continue to take the medication as prescribed until they consult a healthcare professional; healthcare professionals should also continue to follow the prescribing advice on the drug label. ... Post-marketing surveillance data shows that patients receiving saxagliptin have reported experiencing acute pancreatitis. The U.S. Food and Drug Administration (FDA) is evaluating the results of an unpublished study that suggests a potentially increased risk of pancreatitis and precancerous cellular lesions (pancreatic duct metaplasia) in patients with type 2 diabetes treated with incretin analogues (exenatide, liraglutide, sitagliptin, saxagliptin, alogliptin, or linagliptin). These findings are based on the examination of small amounts of pancreatic tissue specimens from patients who died for unknown reasons during treatment with incretin analogues. The FDA has not drawn any new conclusions regarding the safety risks of incretin analogues. The FDA will notify healthcare professionals of its findings and recommendations upon completion of its review or when more information becomes available. The FDA states that clinicians should continue to follow the recommendations in the prescribing information for incretin analogues. The manufacturer states that patients receiving treatment with saxagliptin should be monitored for symptoms of pancreatitis. If pancreatitis is suspected, saxagliptin should be discontinued immediately and appropriate treatment should be initiated. Saxagliptin has not been studied in patients with a history of pancreatitis, so it is unclear whether treatment with saxagliptin in such patients increases the risk of pancreatitis. Post-marketing reports indicate that saxagliptin can cause serious anaphylactic and hypersensitivity reactions (e.g., anaphylactic shock, angioedema, exfoliative dermatitis). These reactions typically occur within the first 3 months of starting treatment; some reactions occur immediately after the first dose. Pharmacodynamics: GLP-1 and GIP levels can increase 2 to 3 times after administration of saxagliptin. Due to the high selectivity of saxagliptin for DPP-4 inhibitors, systemic side effects are relatively few. Saxagliptin inhibits DPP-4 enzyme activity for 24 hours. It also reduces glucagon levels and increases glucose-dependent insulin secretion from pancreatic β-cells. The half-maximal inhibitory concentration (IC50) is 0.5 nmol/L. Saxagliptin does not prolong the QTc interval to a clinically significant degree. Mechanism of Action Saxagliptin is a dipeptidyl peptidase-4 (DPP-4) inhibitor used to treat type 2 diabetes. DPP-4 inhibitors are a class of compounds that act by affecting the body's natural hormone, incretin. Incretin lowers blood glucose by increasing the body's consumption of glucose (primarily through increased pancreatic insulin production) and reducing hepatic glucose production. [Bristol-Myers Squibb Press Release] DPP-4 is a membrane-associated peptidase found in many tissues, lymphocytes, and plasma. DPP-4 has two main mechanisms of action: one is enzymatic function, and the other is binding to adenosine deaminase, which, upon activation, transmits intracellular signals via dimerization. Saxagliptin forms a reversible, histidine-assisted covalent bond between its nitrile group and the S630 hydroxyl oxygen of DPP-4. Inhibition of DPP-4 increases the activity level of glucagon-like peptide-1 (GLP-1), which inhibits glucagon production by pancreatic α cells and promotes insulin production by pancreatic β cells. Type 2 diabetes (T2D) is one of the major risk factors for Alzheimer's disease (AD). Recent studies have found similarities in the molecular mechanisms of the degenerative progression of these two diseases. Drugs that can increase glucagon-like peptide-1 (GLP-1) levels and improve type 2 diabetes, such as dipeptidyl peptidase-4 (DPP-4) inhibitors, have become important candidate disease-modifying drugs for the treatment of Alzheimer's disease (AD). Furthermore, increased endogenous GLP-1 levels can reduce β-amyloid (Aβ) levels and tau protein phosphorylation levels in Alzheimer's disease (AD). This study aimed to investigate the efficacy of the DPP-4 inhibitor saxagliptin in a streptozotocin (STZ)-induced AD rat model. Three months after intracerebral injection of streptozotocin to induce AD, animals were orally administered saxagliptin (0.25, 0.5, and 1 mg/kg) for 60 days. The effects of the DPP-4 inhibitor on hippocampal GLP-1 levels, Aβ load, tau protein phosphorylation, inflammatory markers, and memory retention were evaluated. Results showed that treatment reduced Aβ, tau protein phosphorylation, and inflammatory marker levels, while improving hippocampal GLP-1 levels and memory retention. Saxagliptin exerts a significant therapeutic effect by inhibiting DPP-4, revealing a unique mechanism for Aβ and tau protein clearance—namely, reversing behavioral deficits and pathological changes in Alzheimer's disease (AD) patients by increasing GLP-1 levels. PMID:23603201 Saxagliptin inhibits dipeptidyl peptidase-4 (DPP-4), an enzyme that inactivates the incretin hormone glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP). Saxagliptin and its active metabolite (5-hydroxysaxagliptin) exhibit more selective inhibition of DPP-4 than DPP-8 or DPP-9. Saxagliptin increases circulating levels of GLP-1 and GIP in a glucose-dependent manner. GLP-1 and GIP stimulate insulin secretion from pancreatic β-cells in a glucose-dependent manner (i.e., when blood glucose levels are normal or elevated). GLP-1 also reduces glucagon secretion from pancreatic α-cells, thereby decreasing hepatic glucose production. Saxagliptin can reduce fasting blood glucose levels and decrease glucose load or postprandial blood glucose fluctuations in patients with type 2 diabetes. Background: Type 2 diabetes mellitus (T2DM) is characterized by progressive loss of β-cell function. A promising new approach is the use of oral active dipeptidyl peptidase-4 (DPP-4) inhibitors. However, every new compound for the treatment of type 2 diabetes mellitus (T2DM) must demonstrate its long-term safety, particularly in terms of cardiovascular outcomes. Objective: To conduct a systematic review and meta-analysis of the primary efficacy endpoints and safety profiles of sitagliptin and vildagliptin. Selection criteria, data collection, and analysis: Randomized controlled clinical trials of type 2 diabetes lasting at least 12 weeks were included. Results: Compared with placebo, DPP-4 inhibitors improved glycated hemoglobin A1c (A1c) by 0.7%, but this was not significantly different from other hypoglycemic agents used alone (0.3% improvement in the control group). The overall risk of DPP-4 inhibitors was low, but sitagliptin use was associated with a 34% increased relative risk of all-cause infection (95% confidence interval 10% to 64%, P = 0.004). Data on immune function, health-related quality of life, and diabetic complications were not available. Conclusion: DPP-4 inhibitors are theoretically superior to existing oral hypoglycemic agents, but should currently be limited to individual patient use. Long-term data on cardiovascular outcomes and safety are needed before these new drugs can be widely used. [1] Saxagliptin is a potent and selective dipeptidyl peptidase-4 reversible inhibitor currently being developed for the treatment of type 2 diabetes. It is rapidly absorbed after oral administration and its pharmacokinetic profile is suitable for once-daily dosing. Saxagliptin is metabolized in the body to produce an active metabolite, and both the parent drug and the metabolite are primarily excreted by the kidneys. Saxagliptin reduces the degradation of the incretin hormone glucagon-like peptide-1, thereby enhancing its effect and is associated with improved β-cell function and inhibition of glucagon secretion. Clinical trials lasting up to 24 weeks have shown that saxagliptin monotherapy improves glycemic control and enhances efficacy when used in combination with other oral hypoglycemic agents (metformin, sulfonylureas, thiazolidinediones). Both fasting and postprandial blood glucose concentrations are reduced, resulting in a clinically significant decrease in glycated hemoglobin levels. Because its mechanism of action is glucose-dependent, the risk of hypoglycemia is low. Saxagliptin has been reported to be well tolerated with side effects similar to placebo. It has no effect on body weight, so no dose adjustment is required for age, sex, or liver impairment. Saxagliptin, co-developed by Bristol-Myers Squibb (New York City, NY, USA) and AstraZeneca (Cheshire, UK), is currently under regulatory review. [2] To further elucidate the structure-activity relationship (SAR) of our previously disclosed series of β-quaternary ammonium amino acid-linked l-cis-4,5-methyleneprolyl nitrile dipeptidyl peptidase IV (DPP-IV) inhibitors, we investigated vinyl substitution at the β-position of α-cycloalkyl-substituted glycine. Despite lower systemic exposure, vinyl-substituted compounds showed a prolonged duration of action in an acute rat ex vivo plasma DPP-IV inhibition model. Oxygenated putative metabolites were prepared and demonstrated in a Zucker (fa/fa) rat glucose clearance efficacy model that these metabolites had the same potency and a longer duration of action as their prodrugs. Extending this approach to adamantyl glycine-derived inhibitors has revealed highly potent inhibitors, including the hydroxyadamantyl compound BMS-477118 (saxagliptin), a potent, stable, and long-acting DPP-IV inhibitor currently undergoing clinical trials for the treatment of type 2 diabetes. [3] Type 2 diabetes (T2DM) is a global epidemic with an increasing impact on individuals and healthcare providers. Existing treatments, such as metformin, sulfonylureas, thiazolidinediones, and insulin, are not effective in long-term glycemic control. In addition, most of these treatments have adverse side effects such as weight gain and hypoglycemia. Therefore, exploring new therapeutic targets and therapies is crucial for treating this disease. The incretin pathway, particularly glucagon-like peptide-1 (GLP-1), plays an important pathological role in the development of type 2 diabetes, and therapies targeting the incretin system have emerged in recent years. These drugs can be mainly divided into two categories: GLP-1 agonists/analytes (such as exenatide and liraglutide) and dipeptidyl peptidase-4 (DPP-4; DPP-4 is an enzyme responsible for rapidly inactivating incretin) inhibitors (such as sitagliptin and vildagliptin). Saxagliptin is a novel DPP-4 inhibitor that has recently completed a phase III clinical trial. Saxagliptin is a highly effective and specific DPP-4 inhibitor (compared to other dipeptidyl peptidases) and is administered once daily. Current data indicate that saxagliptin monotherapy or in combination with metformin, glibenclamide, or thiazolidinediones can significantly reduce fasting and postprandial blood glucose and glycated hemoglobin A1c (HbA1c) levels. Saxagliptin is well tolerated, does not increase the risk of hypoglycemia compared to placebo, and may have no effect on weight. Saxagliptin will become one of the many new and effective drugs currently available for the treatment of patients with type 2 diabetes. [4] |
| 分子式 |
C18H25N3O2
|
|---|---|
| 分子量 |
315.4100
|
| 精确质量 |
315.194
|
| CAS号 |
1564266-00-7
|
| 相关CAS号 |
Saxagliptin hydrate;945667-22-1;Saxagliptin hydrochloride;709031-78-7; 361442-04-8; 1073057-20-1 (HCl hydrate); 1073057-33-6 (benzoate hydrate)
|
| PubChem CID |
71752157
|
| 外观&性状 |
Typically exists as solid at room temperature
|
| 密度 |
1.4±0.1 g/cm3
|
| 沸点 |
548.7±35.0 °C at 760 mmHg
|
| 闪点 |
285.6±25.9 °C
|
| 蒸汽压 |
0.0±3.3 mmHg at 25°C
|
| 折射率 |
1.640
|
| LogP |
-0.14
|
| tPSA |
90.4
|
| 氢键供体(HBD)数目 |
2
|
| 氢键受体(HBA)数目 |
4
|
| 可旋转键数目(RBC) |
2
|
| 重原子数目 |
23
|
| 分子复杂度/Complexity |
609
|
| 定义原子立体中心数目 |
6
|
| SMILES |
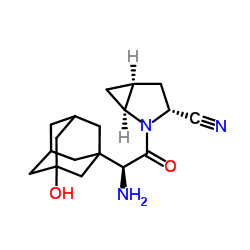
O([H])C12C([H])([H])[C@]3([H])C([H])([H])[C@]([H])(C1([H])[H])C([H])([H])C([C@@]([H])(C(N1[C@@]([H])(C#N)C([H])([H])[C@]4([H])C([H])([H])[C@]14[H])=O)N([H])[H])(C3([H])[H])C2([H])[H]
|
| InChi Key |
QGJUIPDUBHWZPV-BBEOPNRRSA-N
|
| InChi Code |
InChI=1S/C18H25N3O2/c19-8-13-2-12-3-14(12)21(13)16(22)15(20)17-4-10-1-11(5-17)7-18(23,6-10)9-17/h10-15,23H,1-7,9,20H2/t10-,11+,12-,13-,14+,15-,17?,18?/m1/s1
|
| 化学名 |
(1S,3R,5S)-2-[(2S)-2-amino-2-[(5S,7R)-3-hydroxy-1-adamantyl]acetyl]-2-azabicyclo[3.1.0]hexane-3-carbonitrile
|
| 别名 |
1564266-00-7; (2'S,2R,trans)-Saxagliptin; (1S,3R,5S)-2-[(2S)-2-Amino-2-[(5S,7R)-3-hydroxy-1-adamantyl]acetyl]-2-azabicyclo[3.1.0]hexane-3-carbonitrile; 2-Azabicyclo[3.1.0]hexane-3-carbonitrile, 2-[(2S)-2-amino-2-(3-hydroxytricyclo[3.3.1.13,7]dec-1-yl)acetyl]-, (1S,3R,5S)-; (1S,3R,5S)-2-((S)-2-Amino-2-((1r,3R,5R,7S)-3-hydroxyadamantan-1-yl)acetyl)-2-azabicyclo[3.1.0]hexane-3-carbonitrile; (1S,3R,5S)-2-[(2S)-2-Amino-2-(3-hydroxytricyclo[3.3.1.13,7]dec-1-yl)acetyl]-2-azabicyclo[3.1.0]hexane-3-carbonitrile
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples
|
|---|---|
| 溶解度 (体内实验) |
注意: 如下所列的是一些常用的体内动物实验溶解配方,主要用于溶解难溶或不溶于水的产品(水溶度<1 mg/mL)。 建议您先取少量样品进行尝试,如该配方可行,再根据实验需求增加样品量。
注射用配方
注射用配方1: DMSO : Tween 80: Saline = 10 : 5 : 85 (如: 100 μL DMSO → 50 μL Tween 80 → 850 μL Saline)(IP/IV/IM/SC等) *生理盐水/Saline的制备:将0.9g氯化钠/NaCl溶解在100 mL ddH ₂ O中,得到澄清溶液。 注射用配方 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (如: 100 μL DMSO → 400 μL PEG300 → 50 μL Tween 80 → 450 μL Saline) 注射用配方 3: DMSO : Corn oil = 10 : 90 (如: 100 μL DMSO → 900 μL Corn oil) 示例: 以注射用配方 3 (DMSO : Corn oil = 10 : 90) 为例说明, 如果要配制 1 mL 2.5 mg/mL的工作液, 您可以取 100 μL 25 mg/mL 澄清的 DMSO 储备液,加到 900 μL Corn oil/玉米油中, 混合均匀。 View More
注射用配方 4: DMSO : 20% SBE-β-CD in Saline = 10 : 90 [如:100 μL DMSO → 900 μL (20% SBE-β-CD in Saline)] 口服配方
口服配方 1: 悬浮于0.5% CMC Na (羧甲基纤维素钠) 口服配方 2: 悬浮于0.5% Carboxymethyl cellulose (羧甲基纤维素) 示例: 以口服配方 1 (悬浮于 0.5% CMC Na)为例说明, 如果要配制 100 mL 2.5 mg/mL 的工作液, 您可以先取0.5g CMC Na并将其溶解于100mL ddH2O中,得到0.5%CMC-Na澄清溶液;然后将250 mg待测化合物加到100 mL前述 0.5%CMC Na溶液中,得到悬浮液。 View More
口服配方 3: 溶解于 PEG400 (聚乙二醇400) 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.1705 mL | 15.8524 mL | 31.7048 mL | |
| 5 mM | 0.6341 mL | 3.1705 mL | 6.3410 mL | |
| 10 mM | 0.3170 mL | 1.5852 mL | 3.1705 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。