| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| Other Sizes |
|
| 体外研究 (In Vitro) |
藏红花的中枢神经系统作用被认为归因于藏红花酸,这是一种从藏红花素类胡萝卜素中获得的代谢物,已被证明对 NMDA 受体具有显着的亲和力。将细胞线粒体脱氢酶与测试化合物一起孵育 24 小时后,通过 MTT 测定测量 Caco-2 细胞的活性:水醇藏红花提取物(SE,0.5-1 mg/mL)和 crocin-1(250-1000 µM)没有显示出细胞活力的显着变化和阴性结果。这样做是为了确保 Caco-2 细胞的活力在整个运输实验过程中保持不变。细胞活力在 10 µM 浓度下不受藏红花酸影响,但在 40–160 µM 浓度下显着降低 [1]。
|
|---|---|
| 毒性/毒理 (Toxicokinetics/TK) |
相互作用
本研究采用戊四唑(PTZ)诱导小鼠惊厥模型,评估了番红花柱头成分番红醛和番红花素的抗惊厥活性。番红醛(0.15 和 0.35 mL/kg,腹腔注射)可缩短惊厥持续时间,延缓强直性惊厥的发生,并保护小鼠免于死亡。番红花素(200 mg/kg,腹腔注射)未显示抗惊厥活性。/番红醛和番红花素/ 灌胃给予小鼠 125-250.0 mg/kg 体重的番红花柱头 50% 乙醇提取物,可产生镇静作用,并增强巴比妥类药物的镇静作用。番红花柱头乙醇提取物 局部涂抹100 mg/kg体重的95%番红花柱头乙醇提取物可抑制小鼠皮肤癌发生的两阶段起始和发展,延缓乳头状瘤的形成并减少每只小鼠的平均乳头状瘤数量。每日灌胃给予100.0 mg/kg体重的相同提取物,持续30天,可使20-甲基胆蒽诱导的小鼠软组织肉瘤发生率降低10%。灌胃给予100.0 mg/kg体重的番红花柱头乙醇提取物可分别抑制实体Dalton淋巴瘤腹水和肉瘤180肿瘤的生长87%和41%。每周皮下注射400.0 mg/kg体重的番红花素,持续13周,可减缓结肠腺癌的生长,并延长雌性小鼠(而非雄性小鼠)的寿命。/番红花柱头乙醇提取物/ 腹腔注射50 mg/kg体重的95%番红花柱头乙醇提取物,可部分预防顺铂治疗引起的小鼠体重、血红蛋白水平和白细胞计数下降。藏红花乙醇提取物 非人类毒性值 LD50 大鼠(雄性)5.53 mL/kg /藏红花醛/ LD50 大鼠(雄性)1.5 mL/kg /藏红花醛/ LD50 小鼠(雌性)腹腔注射 1.88 mL/kg /藏红花醛/ LD50 小鼠(雄性)腹腔注射 1.48 mL/kg /藏红花醛/ 有关藏红花油(共8种)的更多非人类毒性值(完整)数据,请访问HSDB记录页面。 |
| 参考文献 |
|
| 其他信息 |
番红花酸是一种20碳二羧酸,属于二萜类化合物,也是一种天然类胡萝卜素。它存在于番红花中,曾被用作抗疲劳膳食补充剂。番红花酸具有营养保健、代谢和抗氧化作用。它是一种胡萝卜素酸、二萜类化合物和多不饱和二羧酸。它是藏红花酸(2-)的共轭酸。
维生素A类似物,可增加水溶液(包括血浆)中氧气的扩散率。 据报道,藏红花酸存在于栀子、紫苏以及其他有相关数据的生物体中。 药物适应症 已研究用于治疗癌症/肿瘤(未具体说明)、出血、缺氧、呼吸衰竭和中风。 作用机制 反式藏红花酸钠是一种新型药物,已被证明可增加出血性休克期间的全身耗氧量。它的作用机制是通过增加氧气在血浆中的扩散速率,而不是针对出血性休克的特定症状,因此被认为可作为低氧血症的通用治疗方法。因此,它也可能对治疗呼吸功能不全有益。 治疗用途 EXPL THER:本研究采用20%柠檬酸雾化溶液,在豚鼠体内评估了番红花柱头和花瓣提取物及其成分番红花醛和番红花素的镇咳活性。提取物和药物均采用腹腔注射给药。番红花乙醇提取物(100-800 mg/kg)和番红花醛(0.25-0.75 mL/kg)可减少咳嗽次数。花瓣和番红花素的乙醇提取物和水提取物均未显示镇咳活性。/番红花柱头提取物/ EXPL THER:番红花(Crocus sativus L.)在民间医学中被广泛应用,例如用作抗水肿剂。作者旨在评估藏红花提取物在小鼠体内的镇痛和抗炎活性。他们使用了藏红花(Crocus sativus L.)柱头和花瓣的水浸提取物和乙醇浸提取物。采用热板试验和扭体试验检测了提取物的镇痛活性。使用二甲苯诱导的小鼠耳廓水肿模型研究了提取物对急性炎症的作用。使用福尔马林诱导的大鼠足爪水肿模型评估了提取物对慢性炎症的作用。在热板试验中,腹腔注射两种提取物均未显示出对小鼠的显著镇痛活性。提取物对醋酸诱导的扭体试验表现出镇痛活性。纳洛酮仅部分阻断了柱头水浸提取物的镇痛活性。只有柱头提取物对急性炎症表现出弱至中等的镇痛作用。在慢性炎症中,柱头水浸提取物、乙醇浸提取物以及花瓣乙醇浸提取物均表现出抗炎作用。作者得出结论,藏红花柱头和花瓣的水提物和乙醇提取物具有镇痛作用,以及急性和/或慢性抗炎活性。柱头提取物 实验治疗:每月一次肌注藏红花酸(剂量未指定)给喂食动脉粥样硬化诱导饮食的兔子,可使血清胆固醇浓度降低50%,动脉粥样硬化的严重程度降低约30%。藏红花酸 实验治疗:一项临床试验评估了柱头的抗氧化作用,该试验纳入30名受试者,分为三组:10名健康志愿者、10名冠状动脉疾病患者和10名健康对照者。两个试验组每天两次,连续6周,在100毫升牛奶中加入50毫克藏红花柱头,对照组仅饮用牛奶。与对照组相比,健康志愿者血液样本中的脂蛋白氧化降低了 42.3% (P < 0.001),冠状动脉疾病患者的脂蛋白氧化降低了 37.9% (P < 0.01)。/番红花/ 有关番红花油(共 7 种)的更多治疗用途(完整)数据,请访问 HSDB 记录页面。 药物警告 番红花剂量达到 5.0 克或以上时,可能引起严重不良反应。番红花过量服用(12.0-20.0 克/天)可能致命。/番红花/ 番红花可能诱发子宫收缩,因此孕妇禁用。由于缺乏安全性数据,儿童和哺乳期妇女应将番红花的使用限制在正常食用范围内。出血性疾病患者禁用番红花。圣洁莓(Stigma Croci)会抑制血小板聚集,因此服用抗凝血或抗血小板药物的患者应谨慎使用。 |
| 分子式 |
C20H24O4
|
|---|---|
| 分子量 |
328.4022
|
| 精确质量 |
328.167
|
| CAS号 |
27876-94-4
|
| 相关CAS号 |
Crocetin disodium;591230-99-8;Crocetin meglumine
|
| PubChem CID |
5281232
|
| 外观&性状 |
Yellow to orange solid powder
|
| 密度 |
1.1±0.1 g/cm3
|
| 沸点 |
585.1±23.0 °C at 760 mmHg
|
| 熔点 |
285°
|
| 闪点 |
321.7±19.1 °C
|
| 蒸汽压 |
0.0±3.5 mmHg at 25°C
|
| 折射率 |
1.559
|
| LogP |
4.72
|
| tPSA |
74.6
|
| 氢键供体(HBD)数目 |
2
|
| 氢键受体(HBA)数目 |
4
|
| 可旋转键数目(RBC) |
8
|
| 重原子数目 |
24
|
| 分子复杂度/Complexity |
608
|
| 定义原子立体中心数目 |
0
|
| SMILES |
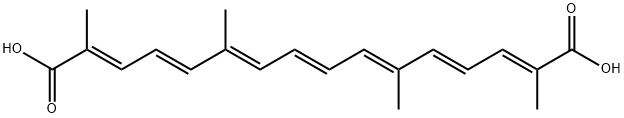
C/C(=C\C=C\C=C(\C=C\C=C(\C(=O)O)/C)/C)/C=C/C=C(/C(=O)O)\C
|
| InChi Key |
PANKHBYNKQNAHN-MQQNZMFNSA-N
|
| InChi Code |
InChI=1S/C20H24O4/c1-15(11-7-13-17(3)19(21)22)9-5-6-10-16(2)12-8-14-18(4)20(23)24/h5-14H,1-4H3,(H,21,22)(H,23,24)/b6-5+,11-7+,12-8+,15-9+,16-10+,17-13+,18-14+
|
| 化学名 |
(2E,4E,6E,8E,10E,12E,14E)-2,6,11,15-tetramethylhexadeca-2,4,6,8,10,12,14-heptaenedioic acid
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month 注意: 该产品在溶液状态不稳定,请现配现用。 |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
DMSO : ~1 mg/mL (~3.05 mM)
|
|---|---|
| 溶解度 (体内实验) |
注意: 如下所列的是一些常用的体内动物实验溶解配方,主要用于溶解难溶或不溶于水的产品(水溶度<1 mg/mL)。 建议您先取少量样品进行尝试,如该配方可行,再根据实验需求增加样品量。
注射用配方
注射用配方1: DMSO : Tween 80: Saline = 10 : 5 : 85 (如: 100 μL DMSO → 50 μL Tween 80 → 850 μL Saline)(IP/IV/IM/SC等) *生理盐水/Saline的制备:将0.9g氯化钠/NaCl溶解在100 mL ddH ₂ O中,得到澄清溶液。 注射用配方 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (如: 100 μL DMSO → 400 μL PEG300 → 50 μL Tween 80 → 450 μL Saline) 注射用配方 3: DMSO : Corn oil = 10 : 90 (如: 100 μL DMSO → 900 μL Corn oil) 示例: 以注射用配方 3 (DMSO : Corn oil = 10 : 90) 为例说明, 如果要配制 1 mL 2.5 mg/mL的工作液, 您可以取 100 μL 25 mg/mL 澄清的 DMSO 储备液,加到 900 μL Corn oil/玉米油中, 混合均匀。 View More
注射用配方 4: DMSO : 20% SBE-β-CD in Saline = 10 : 90 [如:100 μL DMSO → 900 μL (20% SBE-β-CD in Saline)] 口服配方
口服配方 1: 悬浮于0.5% CMC Na (羧甲基纤维素钠) 口服配方 2: 悬浮于0.5% Carboxymethyl cellulose (羧甲基纤维素) 示例: 以口服配方 1 (悬浮于 0.5% CMC Na)为例说明, 如果要配制 100 mL 2.5 mg/mL 的工作液, 您可以先取0.5g CMC Na并将其溶解于100mL ddH2O中,得到0.5%CMC-Na澄清溶液;然后将250 mg待测化合物加到100 mL前述 0.5%CMC Na溶液中,得到悬浮液。 View More
口服配方 3: 溶解于 PEG400 (聚乙二醇400) 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.0451 mL | 15.2253 mL | 30.4507 mL | |
| 5 mM | 0.6090 mL | 3.0451 mL | 6.0901 mL | |
| 10 mM | 0.3045 mL | 1.5225 mL | 3.0451 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT04378920 | Completed | Drug: LEAF-4L6715 | COVID19, Sepsis or Other Causes Acute Respiratory Distress Syndrome |
Institut de cancérologie Strasbourg Europe |
April 14, 2020 | Phase 1 Phase 2 |
|
|