| 规格 | 价格 | |
|---|---|---|
| 500mg | ||
| 1g | ||
| Other Sizes |
| 靶点 |
Ornithine decarboxylase
|
|---|---|
| 体外研究 (In Vitro) |
依氟鸟氨酸是鸟氨酸脱羧酶的一种特异性、不可逆的抑制剂,被认为通过抑制毛囊中的这种酶来减缓头发生长[2]。
采用Franz扩散池对依氟鸟氨酸进行体外渗透研究。当将依氟鸟氨酸乳膏涂抹在用微针预处理过的小鼠皮肤区域时,依氟鸟氨酸的毛发生长抑制活性显著增强,很可能是因为微针产生的微孔允许依氟鸟氨酸渗透到皮肤中,这在体外渗透研究中得到了证实。免疫组织化学数据显示,将依氟鸟氨酸乳膏涂抹在微针预处理的皮肤区域,皮肤和毛囊的细胞增殖也明显受到抑制。[3] |
| 体内研究 (In Vivo) |
过去五十年来唯一被批准用于治疗人类非洲锥虫病的新药是依氟鸟氨酸。它主要用作对 melarsoprol 没有反应的布氏冈比亚锥虫感染的备用药物 [1]。当谈到减少毛发过多的参与者面部毛发的生长时,15% 依氟鸟氨酸霜的效果优于安慰剂。经过 24 周的治疗疗程后,58% 的依氟鸟氨酸患者和 34% 的安慰剂受试者的面部多毛症至少有所改善 [2]。当将依氟鸟氨酸乳膏施用到预先经过微针处理的小鼠皮肤区域时,依氟鸟氨酸的毛发生长抑制活性显着增加[3]。高血压14天后,依氟鸟氨酸治疗缩窄性高血压大鼠导致KCI和去甲肾上腺素收缩强度正常化以及乙酰胆碱松弛[4]。
|
| 酶活实验 |
In vitro permeation of eflornithine hydrochloride through mouse skin/盐酸依氟鸟氨酸体外经小鼠皮肤渗透研究[3]
如前所述,使用Franz扩散池仪完成体外渗透测定(Kumar等人,2012;Kumar et al. 2011;Naguib, Kumar, & Cui 2014)使用C57BL/6小鼠的下背皮肤。收集皮肤前24小时用电动剪发器修剪毛发。采集皮肤,用铝箔包裹,在- 20°C下保存最多一个月,并在需要时使用。在- 20°C下将皮肤冷冻(不使用冷冻保护剂)在文献中是常用的方法,并且这种皮肤样本经常用于渗透性研究(Stahl, Wohlert, & Kietzmann 2012)。Dennerlein等人表明,将新鲜切除的人体皮肤在- 20°C下冷冻和储存长达30天不会影响皮肤的渗透性(Dennerlein等人,2013年)。其他研究人员表明,将人体皮肤包裹在铝箔中并保存在- 26°C时,皮肤可保持其屏障性能长达6个月(Badran, Kuntsche, & Fahr 2009)。去除脂肪层后,将皮肤背朝上置于Franz扩散细胞上。接收器室装有5ml水,用Haake SC 100水循环器(ThermoScientific, Wellington, NH)保持在37°C。如前所述,在将毛发修饰的皮肤安装到Franz扩散池上之前,使用Dermaroller®微针辊进行处理(Kumar等人,2011;Naguib, Kumar, & Cui 2014)。将皮肤样品置于天平的平面上,微针滚轮在皮肤表面沿四个垂直方向滚动,每个方向滚动5次,共滚动20次,施加的压力为350 - 400g,在滚动滚轮的同时使用天平不断测量。皮肤扩散面积为0.64 cm2。供体室以500 μl的水注入盐酸依氟鸟氨酸4 mg,并用副膜覆盖以防止蒸发。在0、1、3、6、8和24 h后,从接收室中取出样品(150 μl),并立即补充新鲜培养基。使用高效液相色谱法对样品进行分析,方法采用先前描述的方法,并进行了修改(Saravanan et al. 2009)。色谱分析采用Agilent 1260 Infinity高效液相色谱仪,配备ZORBAX Eclipse Plus C18 (5 μm, 4.6 × 150 mm)色谱柱,流动相为乙腈-缓冲液(70%:30%,v/v)。将0.68 g磷酸一碱钾溶于1l水中制备缓冲液。流速0.8 ml/min。探测器波长为210 nm。 |
| 细胞实验 |
皮肤组织用福尔马林(10%)缓冲溶液固定24 h,用0.1 M磷酸钠缓冲液(pH 7.4)洗涤,用分级乙醇脱水,石蜡包埋,垂直切片。切片使用苏木精-伊红(H&E)或抗5-溴-2 ' -脱氧尿苷(BrdU)抗体在德克萨斯大学奥斯汀分校戴尔儿科研究所的组织学和组织处理设施进行染色。在安乐死前30分钟,以100 μg/g体重的剂量,在磷酸盐缓冲盐水(PBS, pH 7.4, 10 mM)中腹腔注射BrdU。所有皮肤切片均在Olympus BX53显微镜下检查[3]。
|
| 动物实验 |
In vivo efficacy study was performed in a mouse model by monitoring the re-growth of hair in the lower dorsal skin of mice after the eflornithine cream was applied onto an area pretreated with microneedles. The skin and the hair follicles in the treated area were also examined histologically[3].
Female C57BL/6 mice (8–10 weeks old) were are ideal for examining the physiological actions during different hair cycle phases due to the occurrence of naturally synchronized hair cycles with cyclic pigmentation (Slominski, Paus, & Costantino 1991). Each experimental group was composed of 3–4 mice. Hair in the lower dorsal skin of anesthetized mice was either trimmed using an electric clipper, plucked using GiGi® Honee warm wax as previously described (Xiao et al. 2012), or chemically removed using Nair® lotion. The skin area where the hair was removed was then treated with the eflornithine hydrochloride 13.9% cream (~50 mg per mouse per treatment) using a spatula two times a day in an interval of at least 8 h for a maximum period of 36 days. A group of mice whose hair in the application site was trimmed using a clipper were also treated with the microneedle roller every time before the application of eflornithine cream as previously described (Kumar et al. 2012). Briefly, mice were placed onto the flat surface of a balance, and the microneedle roller was rolled over the marked skin surface, 10 times parallel to mouse length, with an applying pressure of 350–400 g as indicated on the balance. In control groups, the hair in mouse dorsal skin was removed by trimming, plucking, or chemical depilation with Nair®, but the area was not treated with the eflornithine cream. The hair re-growth was evaluated by taking digital photographs of the mouse skin areas for a maximum period of 36 days after the first application of the eflornithine cream. On the last day of the study, animals were euthanized, and skin samples were collected from the treated areas for immunohistochemical studies.[3] |
| 药代性质 (ADME/PK) |
Absorption, Distribution and Excretion
Following oral administration of efornithine, the peak plasma concentration (Cmax) of efornithine is reached at 3.5 hours post-administration (Tmax). Food (high-fat and high-calorie) does not affect the Cmax and AUC (area under the concentration-time curve) of efornithine. Crushing the tablet and adding it to a standard pudding mixture has no effect on the exposure to efornithine (Cmax and AUC6h). Under clinical use conditions, for women with excess facial hair, the mean transdermal absorption of the 13.9% (w/w) efornithine cream formulation, after single or multiple administrations, is less than 1% of the radioactive dose. Clinical use conditions include shaving within 2 hours prior to administration of the radiolabeled dose, as well as other forms of facial hair removal such as shaving, plucking, or tweezing. Steady state is reached within four days with twice-daily administration. Under clinical use conditions, in 10 women with excess facial hair (n=10), applying 0.5 g of cream twice daily (total dose 1.0 g/day; equivalent to 139 mg of anhydrous efornithine hydrochloride) resulted in steady-state Cmax, Ctrough, and AUC12hr of approximately 10 ng/mL, 5 ng/mL, and 92 ng hr/mL, respectively, expressed as anhydrous free base of efornithine hydrochloride. Under steady-state conditions, with twice-daily application of 0.5 g of cream (total dose 1.0 g/day), the dose-normalized peak concentration (Cmax) and daily systemic exposure (AUC) of efornithine are expected to be approximately 100-fold and 60-fold lower, respectively, than with a once-daily oral dose of 370 mg. This compound is not metabolized and is primarily excreted unchanged in the urine. The volume of distribution (Vz/F) of efornithine is 24.3 L. The clearance (CL/F) of efornithine is 5.3 L/h. Under clinical use conditions, in female patients with facial hirsutism, the mean transdermal absorption of efornithine after a single or multiple doses of the 13.9% (w/w) cream formulation was less than 1% of the radioactive dose. Clinical use conditions included shaving within 2 hours prior to application of the radiolabeled drug. Apart from other methods of facial hair removal such as cutting, plucking, or tweezing, under clinical use conditions, in women (n=10) with excess facial hair, after twice-daily application of 0.5 g of the cream (total dose 1.0 g/day; equivalent to 139 mg of anhydrous efornithine hydrochloride), the steady-state Cmax, Ctrough, and AUC12hr, expressed as free base of anhydrous efornithine hydrochloride, were approximately 10 ng/mL, 5 ng/mL, and 92 ng/mL, respectively. At steady state, compared to 370 mg daily, the dose-normalized peak concentration (Cmax) and daily systemic exposure (AUC) of efflunitine were estimated to be reduced by approximately 100-fold and 60-fold, respectively, by twice-daily application of 0.5 g of cream (total dose 1.0 g/day). Oral administration once daily. Eflunitine is not metabolized and is excreted unchanged in the urine. For more complete data on the absorption, distribution, and excretion of efflunitine (8 metabolites), please visit the HSDB record page. Metabolism/Metabolites This compound is not metabolized and is primarily excreted unchanged in the urine. Biological Half-Life The terminal plasma elimination half-life of efflunitine is 3.5 hours, and the apparent steady-state plasma half-life is approximately 8 hours. The apparent steady-state plasma t1/2 of efflunitine is approximately 8 hours. |
| 毒性/毒理 (Toxicokinetics/TK) |
Effects During Pregnancy and Lactation
◉ Overview of Lactation Use Mothers received daily intravenous infusions of 400 mg/kg efflunitine for 7 days without any serious adverse effects on breastfed infants. Topical efflunitine is poorly absorbed and therefore unlikely to enter the infant's bloodstream, and is unlikely to cause any adverse effects on breastfed infants. ◉ Effects on Breastfed Infants In the Democratic Republic of Congo, a cohort study of 33 infants followed hospitalized mothers taking nifurulimus who were breastfeeding (feeding extent not specified). 30 mothers completed 30 doses of oral nifurulimus (15 mg/kg/day), and all mothers received 14 intravenous infusions of efflunitine (400 mg/kg/day) for 7 days for the treatment of human African trypanosomiasis (sleeping sickness). On average, breastfeeding mothers were concurrently taking four other medications, including amoxicillin, ciprofloxacin, metronidazole, sulfamethoxazole/trimethoprim, aspirin, and diclofenac (1 case each); hydrocortisone, promethazine, and quinine (2 cases each); levamisole (6 cases); sulfadoxine-pyrimethamine (8 cases); aminopyrine (13 cases); acetaminophen (16 cases); and mebendazole (17 cases). No serious adverse events were reported in any of the breastfed infants. ◉ Effects on lactation and breast milk As of the revision date, no relevant published information was found. Protein bindingEffornithine does not specifically bind to human plasma proteins. |
| 参考文献 | |
| 其他信息 |
Therapeutic Uses
Antitumor drug; enzyme inhibitor; trypanosome killer. Eflunominosine is indicated for reducing unwanted facial hair in women. Eflunominosine has only been studied in affected individuals in the face and below the chin, adjacent to the affected area. Use should be limited to these affected areas. /US product label contains/ /Experimental Therapy:/ D,L-α-difluoromethylornithine (DFMO) was synthesized over 20 years ago. It was hoped that this enzyme-activated, irreversible inhibitor of ornithine decarboxylase (the first enzyme in polyamine synthesis) would be effective in treating proliferative diseases, including cancer and/or infectious diseases. Studies have found that DFMO typically has cytotoxic effects on mammalian cells and tissues, limiting its therapeutic efficacy. High doses of DFMO have been found to cause therapeutically limiting (but reversible) ototoxicity. This side effect, along with its weak therapeutic activity, has led many clinicians to lose interest in further developing DFMO as a cancer treatment. However, subsequent studies have shown that DFMO can inhibit carcinogen-induced cancer development in various rodent models, leading to increasing interest in developing this compound as a preventative agent. In recent years, the theoretical basis for inhibiting ornithine decarboxylase as a cancer chemopreventive agent has been strengthened, as studies have shown that in certain cell/tissue types, this enzyme can be transactivated by the c-myc oncogene and synergistically act with the ras oncogene to promote malignant transformation of epithelial tissues. Recent dose-decrease-designed clinical chemopreventive trials have demonstrated that DFMO can be administered at low doses over a long period, inhibiting polyamine levels in the gastrointestinal tract and other epithelial tissues without causing detectable hearing loss or other side effects. Ongoing clinical chemopreventive trials are investigating the efficacy of DFMO in inhibiting surrogate endpoint biomarkers of cancer development (e.g., recurrence of colonic polyps) in high-risk populations for certain epithelial cancers (including colon cancer, esophageal cancer, breast cancer, skin cancer, and prostate cancer). Melararsine is often the first-line treatment for Gambian sleeping sickness caused by trypanosomiasis, but prognosis is generally poor if treatment fails. The polyamine synthesis inhibitor efflunitine (difluoromethylornitine, DFMO) has emerged as an alternative therapy. In rural Zaire, an open-label trial treated 207 patients with advanced Gambian trypanosomiasis sleeping sickness using three different DFMO regimens. During treatment, in 87 patients who had trypanosomes detected in their cerebrospinal fluid (CSF) before DFMO administration, the trypanosomes disappeared completely from their CSF, and the CSF white blood cell count decreased dramatically from a mean of 186/µL to 21/µL. 152 patients were followed up for at least one year after DFMO treatment, with only 13 (9%) experiencing relapse. Treatment failure was more common in children under 12 years of age, patients receiving only oral DFMO, and patients using DFMO as initial treatment for newly diagnosed trypanosomiasis. Toxicity was acceptable. Only 4 patients died during or shortly after treatment. Bone marrow suppression leading to anemia (43%) or leukopenia (53%) was relatively common but had minimal impact. This open-label trial suggests that DFMO is comparable in efficacy to melarsone and may have lower toxicity. Due to economic and logistical reasons, DFMO may not be the first-line treatment in rural Africa, but it has a curative effect on the vast majority of patients who relapse after melarisol treatment. ...A polyamine biosynthesis inhibitor, efflunitine (chemical name: DL-α-difluoromethylornitine, supplied as hydrochloride monohydrate), was used to treat a 3.5-year-old child newly diagnosed with severe trypanosomiasis, with an infection history of two years prior in Zaire or Congo. The treatment regimen was: continuous intravenous infusion of efflunitine 300 to 400 mg/kg/day for 25 days; followed by oral efflunitine 300 mg/kg/day, divided into four equal doses, for 17 days. The child's recovery was significant; parasites were cleared from the blood and cerebrospinal fluid within the first week. Cerebrospinal fluid cytosis completely disappeared. Generalized lymphadenopathy and fever gradually subsided. Symptoms such as severe ataxia, inability to walk or voluntarily change posture, significant language regression, and somnolence improved during and after treatment. The child tolerated the drug well; the only observed adverse reaction was transient thrombocytopenia occurring in the fourth week of treatment. Eflunominosine is a safe and effective treatment for central nervous system involvement in this child's trypanosomiasis. Drug Warnings Contraindications include known hypersensitivity to efnominosine or any component of the formulation. If an allergic reaction occurs, discontinue use of efnominosine hydrochloride cream. Transient tingling or burning sensation may occur if applied to broken or abraded skin. Skin irritation may occur in susceptible patients or patients using doses higher than recommended. No persistent laboratory abnormalities have been found associated with efnominosine. In an open-label study, elevated transaminase levels were observed in some patients; however, the clinical significance of these findings is unclear. For more complete data on drug warnings for efflunitine (11 in total), please visit the HSDB record page. Pharmacodynamics Efluflunitine restores the balance of the LIN28/Let-7 metabolic pathway by inhibiting polyamine synthesis and reducing the expression of the oncogenic driver genes MYCN and LIN28B in MYCN-amplified neuroblastoma. This pathway is involved in the regulation of cancer stem cells and glycolytic metabolism. In vitro experiments have shown that efflunitine can induce senescence in MYCN-amplified and non-amplified neuroblastoma cells and inhibit neurosphere formation, suggesting its cytoseptic effect. In mice injected with limited dilutions of MYCN-amplified neuroblastoma cells, treatment with efflunitine can prevent or delay tumor formation. Furthermore, polyamines are also involved in keratin synthesis; inhibiting polyamines can reduce the proliferation of hair follicle stromal cells, thereby inhibiting the anagen phase of hair growth. |
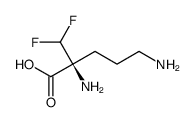
| 分子式 |
C6H12N2O2F2
|
|---|---|
| 分子量 |
182.16848
|
| 精确质量 |
182.087
|
| CAS号 |
67037-37-0
|
| 相关CAS号 |
Eflornithine;70052-12-9;L-Eflornithine monohydrochloride;69955-42-6;Eflornithine hydrochloride;68278-23-9;L-Eflornithine;66640-93-5; 96020-91-6 (HCl hydrate) 68278-23-9 (HCl); 70050-55-4 (R-isomer); 66640-93-5 (L-isomer); 69955-42-6 (S-isomer);
|
| PubChem CID |
3009
|
| 外观&性状 |
White to light yellow solid powder
|
| 密度 |
1.293g/cm3
|
| 沸点 |
347ºC at 760 mmHg
|
| 闪点 |
163.7ºC
|
| 折射率 |
1.462
|
| LogP |
1.173
|
| tPSA |
89.34
|
| 氢键供体(HBD)数目 |
3
|
| 氢键受体(HBA)数目 |
6
|
| 可旋转键数目(RBC) |
5
|
| 重原子数目 |
12
|
| 分子复杂度/Complexity |
166
|
| 定义原子立体中心数目 |
0
|
| InChi Key |
VLCYCQAOQCDTCN-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C6H12F2N2O2/c7-4(8)6(10,5(11)12)2-1-3-9/h4H,1-3,9-10H2,(H,11,12)
|
| 化学名 |
2,5-diamino-2-(difluoromethyl)pentanoic acid
|
| 别名 |
EFLORNITHINE; 70052-12-9; dfmo; Difluoromethylornithine; Ornidyl; 2-(Difluoromethyl)ornithine; 2,5-diamino-2-(difluoromethyl)pentanoic acid; ...; 67037-37-0;
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
Typically soluble in DMSO (e.g. 10 mM)
|
|---|---|
| 溶解度 (体内实验) |
注意: 如下所列的是一些常用的体内动物实验溶解配方,主要用于溶解难溶或不溶于水的产品(水溶度<1 mg/mL)。 建议您先取少量样品进行尝试,如该配方可行,再根据实验需求增加样品量。
注射用配方
注射用配方1: DMSO : Tween 80: Saline = 10 : 5 : 85 (如: 100 μL DMSO → 50 μL Tween 80 → 850 μL Saline)(IP/IV/IM/SC等) *生理盐水/Saline的制备:将0.9g氯化钠/NaCl溶解在100 mL ddH ₂ O中,得到澄清溶液。 注射用配方 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (如: 100 μL DMSO → 400 μL PEG300 → 50 μL Tween 80 → 450 μL Saline) 注射用配方 3: DMSO : Corn oil = 10 : 90 (如: 100 μL DMSO → 900 μL Corn oil) 示例: 以注射用配方 3 (DMSO : Corn oil = 10 : 90) 为例说明, 如果要配制 1 mL 2.5 mg/mL的工作液, 您可以取 100 μL 25 mg/mL 澄清的 DMSO 储备液,加到 900 μL Corn oil/玉米油中, 混合均匀。 View More
注射用配方 4: DMSO : 20% SBE-β-CD in Saline = 10 : 90 [如:100 μL DMSO → 900 μL (20% SBE-β-CD in Saline)] 口服配方
口服配方 1: 悬浮于0.5% CMC Na (羧甲基纤维素钠) 口服配方 2: 悬浮于0.5% Carboxymethyl cellulose (羧甲基纤维素) 示例: 以口服配方 1 (悬浮于 0.5% CMC Na)为例说明, 如果要配制 100 mL 2.5 mg/mL 的工作液, 您可以先取0.5g CMC Na并将其溶解于100mL ddH2O中,得到0.5%CMC-Na澄清溶液;然后将250 mg待测化合物加到100 mL前述 0.5%CMC Na溶液中,得到悬浮液。 View More
口服配方 3: 溶解于 PEG400 (聚乙二醇400) 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 5.4894 mL | 27.4469 mL | 54.8938 mL | |
| 5 mM | 1.0979 mL | 5.4894 mL | 10.9788 mL | |
| 10 mM | 0.5489 mL | 2.7447 mL | 5.4894 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。